A Note to the Reader: This article requires an attention span longer than a fifteen-second reel. We are stepping out of the shallow end of lifestyle tips and into the deep waters of neurobiology and the systemic failures of modern research. So If you are ready to examine the flaws in the science itself, proceed with caution
The Era of Sleep Optimization
Today, the standard for a healthy life is measured in hours: specifically, the eight to nine hours of consecutive sleep demanded by modern health educators. This is not just a lifestyle trend; it is a clinical requirement built on decades of laboratory research. By using tools like polysomnography to map brain waves and muscle tone, scientists have established a rigid “gold standard” for rest. We have turned the bedroom into a hub for optimization, yet despite our intense focus on the science of sleep, sleep disorders and general exhaustion are increasing across the developed world.
Current recommendations rely on data from subjects whose biology is shaped by modern stressors, specifically chronic caffeine intake and glucose-heavy diets. These factors inevitably shift how the brain moves through sleep stages. To accept these laboratory results as a universal biological mandate is akin to studying an inflamed heart and claiming it represents the healthy human norm. We are essentially building a science around a “ruined” subject, documenting the symptoms of a modern lifestyle rather than the true, unadulterated architecture of human rest.
Furthermore, the laboratory environment itself introduces a critical contradiction. In science, the observer effect dictates that the way you monitor a biological process should not change the process itself. Real sleep requires the nervous system to switch off and feel secure, yet the laboratory environment, with its technical equipment and physical wires attached to the scalp and face, makes this impossible. By placing a person in such an unnatural setting, we are attempting to measure the pure biological mandate of sleep in a place that fundamentally prevents it.
This leads to a fundamental question for sleep science: Can we truly define “normal” sleep by studying a body that is constantly being manipulated and in an environment that alters the very thing we study?
The Vigilant Brain: The First Night Effect and the Ancient Survival Approach
Researchers have long known about the First Night Effect. This is the observation that a person’s sleep architecture changes significantly during their initial night in a laboratory. Using brain imaging and EEG monitoring, scientists have found that in a new environment, the left side of the brain maintains heightened activity. This is an ancient survival mechanism called unihemispheric slow-wave sleep. It is the same vigilance pattern seen in dolphins, seals, and migratory birds, where one half of the brain keeps watch while the other rests. The human brain, when sleeping in an unfamiliar place, appears to use a modified version of this survival tactic.
When the brain is in this half-alert state, several essential processes are modified. The glymphatic system, which acts as the brain’s waste clearance, requires both sides of the brain to work together in deep sleep to function properly. Without this bilateral synchronization, the cleaning process is less efficient. Additionally, Stage 4 deep sleep becomes fragmented, and the process of saving memories can be compromised because the brain is busy staying alert. Standard practice in sleep research tries to account for this by throwing out the data from the first night, assuming the brain relaxes on the second night.
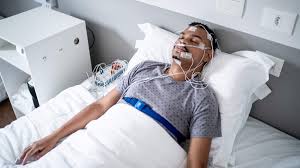
But biology suggests a more complex reality. The brain’s assessment of safety is not just about being familiar with a room. It involves sensory inputs like air movement, temperature, and critically, the absence of physical constraints or foreign objects on the body. Polysomnographic sleep studies require attaching multiple electrodes to the subject’s scalp, face, chest, and sometimes limbs. A standard setup might include 19 scalp electrodes for EEG, 2 eye movement leads, 3 leads on the chin and legs to measure muscle tone, 2 heart rate leads on the chest, respiratory belts around the chest and abdomen, a nasal airflow sensor, an oxygen probe on the finger, and a position sensor. These attachments are secured with conductive paste and adhesive, and the wires are bundled and connected to amplifiers near the bed.
From a neurobiological perspective, sleep represents the most vulnerable state an organism can occupy. The transition into sleep requires the nervous system to assess that the environment is safe enough to permit decreased vigilance. For hundreds of thousands of years, the tactile signal for safety during sleep was the absence of physical constraint or foreign attachments to the body. The presence of electrodes introduces a continuous, novel tactile stimulus. While subjects typically report getting used to the sensation, the deactivation of threat-monitoring systems is a distinct process.
The part of the brain that regulates arousal, known as the ascending reticular activating system, receives continuous input from the body’s sensors. Even when conscious awareness of the electrodes diminishes, the deeper parts of the brain continue to process this novel information. This physical interference has specific physiological consequences. The deepest stages of sleep are associated with a significant drop in core body temperature. This shift requires the hypothalamus to assess that the body is in a safe, stable position. The presence of physical attachments provides contradictory information to this ancient assessment system.
Additionally, the transition of the nervous system into deep sleep involves the withdrawal of the adrenal stress response. Physical constraints historically signaled potential threats, like being trapped or entangled, which maintains a baseline level of stress response in the body. While this response is subtle and often below the threshold of conscious awareness, its presence can be detected in heart rate variability and cortisol levels.
The question, then, is not whether subjects can sleep in laboratory conditions, clearly they can, but whether the sleep architecture observed in these conditions represents the full expression of natural human rest, or merely sleep as it occurs under conditions of subtle, persistent arousal. This distinction is critical to the very foundation of sleep recommendations. If the physical constraints of the laboratory prevent the body from reaching its peak restorative state, the brain is forced to compensate by extending the duration of sleep to achieve a baseline level of recovery. Consequently, the “eight-hour” standard may not be a biological requirement at all, but rather a byproduct of an inefficient environment. If a subject can never reach true, unadulterated restorative sleep while tethered to sensors, then the data used to decide how many hours a human “needs” is fundamentally compromised and questionable at best.
Metabolic State and Sleep Architecture
A second fundamental consideration in interpreting sleep research involves the metabolic state of the subjects. Sleep duration is not a fixed biological constant. Instead, it represents the time the body needs to complete necessary maintenance and repair. If subjects enter sleep with heavy metabolic burdens, their sleep requirements will naturally change.
Most research subjects consume diets high in refined carbohydrates and processed seed oils. In a standard diet, 50% to 60% of calories come from carbohydrates. When these are consumed in the hours before bed, the body must perform several metabolic tasks before it can fully transition into repair mode. Following a high-carbohydrate meal, blood glucose rises and triggers insulin secretion. It typically takes two to four hours for glucose and insulin to return to baseline. During this time, the liver is busy processing the glucose load, converting it to glycogen or, if stores are full, into fatty acids. Simultaneously, the body must process omega-6 polyunsaturated fatty acids from seed oils. These fats are unstable and susceptible to lipid peroxidation, which generates reactive oxygen species that the antioxidant system must neutralize.
The relevance to sleep research is clear: if a subject enters sleep while these processes are ongoing, the body must split its resources between metabolic cleanup and tissue repair. Sleep studies that measure time in various stages are, in part, simply measuring the time required to finish these cleanup operations. Research on fasted versus fed sleep supports this. Subjects who eat within two hours of sleep show longer wait times to reach deep sleep and lower overall efficiency compared to those who fasted for six or more hours. Fed subjects require more total sleep time to feel restored because their bodies were busy with digestion.
Falling into deep sleep is also closely linked to a drop in core body temperature. This temperature decline is functionally necessary for sleep-dependent processes. For example, the glymphatic clearance system, which removes waste from the brain, operates most efficiently when brain temperature is reduced. Similarly, the secretion of growth hormone peaks during deep sleep and is highly sensitive to temperature. Digestion is a thermogenic process. The thermic effect of food can raise core body temperature by 0.3 to 0.5°C and persist for up to six hours. Protein has the highest heating effect, followed by carbohydrates and fats.
When subjects sleep while actively digesting, they are trying to initiate deep sleep processes while their core temperature remains too high. This creates a physiological conflict. The brain may cycle through sleep stages, but the depth and efficiency of those stages are reduced. Because lab protocols rarely require extended fasting, they systematically overestimate how much sleep a person needs when digestion is not a factor.
Another factor is the fuel the body uses during the night. People on high-carbohydrate diets rely primarily on glucose, stored as glycogen. However, these stores are finite. During sleep, the body eventually depletes its glycogen reserves and must switch to alternative fuel sources. For those adapted to high-carbohydrate diets, this transition is metabolically demanding. As glycogen runs low, the body must increase cortisol to produce new glucose and begin burning fat. This shift can trigger arousal as the body responds to perceived low blood sugar. Studies show that people on high-carbohydrate diets often experience more frequent awakenings in the second half of the night when glycogen depletion is at its peak.
In contrast, individuals adapted to burning fat show different patterns. Since they already rely on fat oxidation, the transition during sleep is less dramatic. Fat stores are vast compared to glycogen, allowing fat oxidation to proceed steadily throughout the night without metabolic stress. Again, if a significant portion of sleep is spent on metabolic cleanup rather than fundamental repair, then sleep requirements likely vary based on a person’s metabolic state.
Caffeine and the Adenosine System
Perhaps the most significant flaw in contemporary sleep research is the near-universal consumption of caffeine. Approximately 85% to 90% of adults in developed nations consume caffeine daily. To understand why this ruins sleep data, we must look at how sleep pressure builds and how caffeine sabotages that process.
The biological drive to sleep is governed by adenosine, a chemical that builds up in the brain the longer we stay awake. Adenosine is a byproduct of cellular energy metabolism. As your cells break down ATP for energy, adenosine is released as waste. This chemical acts as an elegant biological timer. It binds to specific receptors (A1 and A2A) in the brain, which then inhibit arousal-promoting chemicals like dopamine and norepinephrine. This creates a progressive increase in sleep pressure throughout the day, ensuring that sleep occurs often enough to clear out metabolic waste.
Caffeine’s primary job is to act as an adenosine antagonist. Because the caffeine molecule is shaped similarly to adenosine, it can fit into those same receptors. However, caffeine does not activate them; it simply blocks them. This is not a minor interference. When caffeine occupies these receptors, the sleep pressure signal is silenced. The adenosine is still there, continuing to pile up, but the brain cannot detect it. This is exactly like placing a piece of tape over a car’s fuel gauge. The tank continues to empty, but the driver sees no change.
The half-life of caffeine averages five to six hours, though this varies based on genetics and CYP1A2 enzyme activity. This means that caffeine consumed at noon is still at 25% concentration at midnight. Even a morning cup of coffee can disrupt evening adenosine signaling for many people.
The brain is a homeostatic organ, meaning it actively works to maintain a stable state. When adenosine receptors are chronically blocked by caffeine, the brain responds by creating more receptors to catch whatever adenosine it can. This process is called upregulation. Research using PET scans has shown that regular caffeine consumers have significantly higher densities of adenosine receptors than non-consumers. This means the physical architecture of the brain, the very gray matter itself, is fundamentally different in caffeine users.
A brain with these extra receptors is hypersensitive; it requires much stronger sleep signals just to initiate rest. It takes weeks or months, not days, for this brain structure to return to normal. Yet, most sleep studies only ask subjects to abstain from caffeine for 12 to 24 hours. Researchers are therefore measuring the sleep of individuals whose brains are structurally altered and likely still contain residual caffeine. The data collected reflects a withdrawal state or an altered receptor state, not the baseline human condition.
Caffeine also has a specific, damaging impact on REM (Rapid Eye Movement) sleep, the stage responsible for memory consolidation and emotional processing. By increasing dopamine signaling, caffeine interferes with the brain’s ability to trigger and maintain REM sleep. In lab studies of habitual caffeine users, researchers often observe a delay in REM sleep, sometimes up to two hours after falling asleep. The standard scientific interpretation is that REM sleep is “fragile” or “difficult to achieve.” However, a more accurate interpretation is that this suppression is a specific side effect of the caffeine-altered brain. In people who have been caffeine-free for long periods, REM sleep begins much earlier and lasts longer.
By studying a caffeine-dependent population, science has mistakenly labeled a “medicated” sleep architecture as the human norm. The question is not whether sleep studies acknowledge caffeine use; most do. The question is whether short-term abstinence is sufficient to normalize the sleep of individuals whose brains have been structurally altered by chronic caffeine consumption. When we study people whose biology has been fundamentally altered, we are not conducting a study of human biology. Instead, we are studying a pathology. We are merely documenting how a broken system attempts to patch itself. To take these findings and call them “biological truths” is like prescribing a specific diet for a diabetic and then claiming it is the universal standard for all human health. We are not measuring the natural requirements of the species; we are measuring the survival tactics of a chronically medicated one.
Light, Circadian Biology, and the Modern Environment
The circadian system is one of the most fundamental parts of human biology. This internal clock coordinates almost every physiological process, from hormone secretion to immune function. The master clock resides in the suprachiasmatic nucleus (SCN) of the hypothalamus, and it is synchronized to the day-night cycle through light hitting the eyes.
The discovery of melanopsin cells in the eye changed our understanding of the brain. Unlike the cells that help us see images, these specialized cells respond primarily to blue light. They send signals directly to the SCN, telling the body whether it is day or night. Natural sunlight provides a full spectrum of light but is rich in blue wavelengths during midday. As the sun sets, the spectrum shifts toward orange and red, and blue light intensity drops. This shift gives the brain reliable information about the time of day.
Modern humans, however, experience a light pattern that is a biological disaster. LED lights, smartphones, and computer screens emit high levels of blue light. We now spend our days in dim indoor light and our evenings under bright, blue-rich artificial light. This is the exact inverse of the natural pattern that shaped our species.
Melatonin is often called the “hormone of darkness,” but it is more than a sleep aid; it is the biochemical signal for “biological night.” Its production begins in the pineal gland when the eyes stop detecting blue light. Exposure to blue light in the evening can suppress melatonin secretion by 50% to 85%. This is not just a delay; it is a total disruption of the body’s signaling system. When light suppresses melatonin, the body enters a state of biological ambiguity. The clock in the brain thinks it is daytime because of the light, but the actual time is night.
The consequence for sleep research is that laboratory subjects arrive for study with their melatonin already suppressed. Their biology thinks it is much earlier than the clock says it is. When these subjects try to sleep, they experience prolonged “sleep onset latency,” they lie awake for 30 to 60 minutes before finally falling asleep. This is not because human sleep naturally requires a long transition period; it is because their circadian system has not yet given the signal for rest. Researchers factor this struggle into their estimates of “normal” sleep. However, this represents the sleep of a circadian-disrupted individual, not the baseline of human physiology.
The human circadian system is not a perfect 24-hour clock; it is roughly 24.2 to 24.5 hours. To stay synchronized, the internal clock must be reset every single morning by bright morning light. Sunlight in the first hours after waking advances the circadian phase, shifting the entire rhythm earlier. This morning light also sets a biological timer for the evening. Sleep laboratories, however, are typically windowless environments. While this gives researchers experimental control, it means subjects are disconnected from the natural cycle that anchors their rhythms. Sleep measured in this “unanchored” condition is not a true representation of sleep as it occurs when a body is properly synced to the solar day.
Thermal Environment and Sleep Physiology
A final consideration that receives surprisingly little attention is the thermal environment. Sleep laboratories maintain a constant ambient temperature, typically around 20-22°C (68-72°F). This stability is maintained for experimental standardization, but it removes a signal fundamental to human sleep. Human sleep is characterized by a significant decrease in core body temperature, typically dropping by 0.5-1.0°C. This decrease is functionally necessary for several sleep-dependent processes, including the optimal function of the glymphatic system.
Throughout human evolution, sleep occurred in environments with significant thermal variation. As the sun set, ambient temperature decreased. This environmental cooling assisted the body’s physiological cooling, meaning the hypothalamus didn’t have to work as hard to initiate the temperature drop required for deep sleep. In a thermally stable environment, the body must generate the entire temperature drop through internal changes alone. This requires additional time or effort to achieve the same depth of cooling.
Furthermore, laboratory air quality differs from natural night air. Natural air contains negative ions and subtle movement patterns that influence respiratory depth and efficiency. Some research suggests that negative ion exposure enhances parasympathetic nervous system activity, the “rest and digest” state that predominates during deep sleep. Shallow breathing, which may occur in static air environments, could theoretically reduce the efficiency of glymphatic clearance. If the body requires additional time to generate physiological cooling without environmental assistance, then laboratory sleep duration might overestimate the sleep needs of individuals sleeping in more natural conditions.
Conclusion: Toward a New Architecture of Rest
The current landscape of sleep science is built upon a fundamental error. By treating the laboratory-bound, caffeine-dependent, and metabolically burdened individual as the “gold standard” for human biology, we have created a science of pathology rather than health. We have spent decades documenting how a compromised body manages to shut down, and we have mistakenly called the results a universal requirement.
Our eight-to-nine-hour sleep mandate is not a biological constant; it is a clinical observation of a body under stress. When we account for the observer effect, metabolic cleanup, and circadian disruption, the “normal” sleep duration begins to look like a symptom of inefficiency. We are not measuring the true architecture of human rest; we are measuring the time it takes for a modern human to recover from biological insults.
Sleep is a dynamic process that varies with metabolic health, the environment, and internal chemistry. A person who is optimized, caffeine-free, and anchored to the natural light-dark cycle may require significantly less sleep than the laboratory “average” because their body is more efficient at repair. Sleep science must stop trying to patch a broken system and start studying the baseline. The goal of a healthy life is not to hit a numerical target, but to cultivate a biological state where sleep is so efficient that it becomes a seamless extension of our health. The eight-hour sleep is not a requirement for being human; it is a requirement for surviving the modern world. It is time we built a science that understands the difference.
References
I. Evolutionary Sleep & The Flaws of Modern Research
Books:
- “Evolution of Sleep: Phylogenetic and Functional Perspectives” by Patrick McNamara, Charles L. Nunn, and Robert A. Barton. (Cambridge University Press). Examines why human sleep evolved the way it did and why modern clinical settings fail to replicate natural conditions.
- “Shining Evolutionary Light on Human Sleep and Sleep Disorders” (Research Compendium) by Charles L. Nunn and David R. Samson. Explores the gap between how we sleep now and how we slept throughout human history.
- “At Day’s Close: Night in Times Past” by A. Roger Ekirch. The primary historical reference for “first” and “second” sleep, proving that the consolidated eight-hour block is a modern invention.
Research Papers:
- [Night watch in one brain hemisphere during sleep associated with the first-night effect in humans] (Tamaki et al., 2016, Current Biology). Link
- Direct evidence for unihemispheric vigilance (the ancient survival approach) in laboratory settings.
- [Surveillance During REM Sleep for the First-Night Effect] (Tamaki et al., 2019, Frontiers in Neuroscience). Link
- Confirms that the brain remains alert to “deviant” sounds in new environments even during REM.
II. Caffeine & The Medicated Brain (Adenosine System)
Research Papers:
- [Sleep Deprivation Increases A1 Adenosine Receptor Binding in the Human Brain] (Elmenhorst et al., 2007, Journal of Neuroscience). Link
- The definitive study on how the brain creates more receptors (upregulation) to handle chronic blockage by caffeine.
- [Adenosine, caffeine, and sleep–wake regulation: state of the science] (Fredholm et al., 2022). Link
- A review of how caffeine fundamentally reconfigures the adenosinergic system.
- [Caffeine Effects on Sleep Taken 0, 3, or 6 Hours before Bedtime] (Drake et al., 2013). Link
- Shows that even caffeine consumed six hours before bed significantly reduces total sleep time and quality.
III. Metabolic Burden & The Efficiency of Repair
Research Papers:
- [The Associations Between Sleep Duration and Various Metabolic Health Indices] (NHANES Data Analysis, 2025). Link
- Details the U-shaped relationship between sleep and metabolic health, supporting the idea that “Standard” sleep reflects metabolic burden.
- [The links between sleep duration, obesity and type 2 diabetes mellitus] (Journal of Endocrinology, 2025). Link
- Explores the bi-directional relationship where metabolic disease “ruins” the subject’s sleep architecture.
- [Interactions between sleep and metabolism] (Sharma & Kavuru, 2010). Link
- Describes how glucose and insulin levels interfere with deep sleep and growth hormone release.
IV. Environmental Disruption (Light & Temperature)
Research Papers:
- [Impacts of Blue Light Exposure From Electronic Devices on Circadian Rhythm] (ResearchGate Review, 2024). Link
- Evidence on how evening blue light causes a 1.5-hour delay in melatonin onset.
- [Effects of thermal environment on human sleep and thermoregulation] (Muzet et al., 2013). Link
- Confirms that human sleep is sensitive to ambient temperature and requires a core body temperature drop to be efficient.
- [Thermal environment and sleep quality: A review] (ResearchGate, 2025). Link
- Proves that thermal adaptive behaviors (like cooling) significantly improve sleep efficiency.
V. Orthosomnia & The Performance Paradox
Research Papers:
- [Orthosomnia: Are Some Patients Taking the Quantified Self Too Far?] (Baron et al., 2017, Journal of Clinical Sleep Medicine). Link
- The landmark paper that defined “Orthosomnia” and the anxiety caused by tracking “perfect” sleep.
- [A qualitative study of sleep trackers usage: evidence of orthosomnia] (CIEHF, 2020). Link
- Shows that users felt more alert during a week without a tracking device, highlighting the performance anxiety paradox.

Leave a comment